
Trukera Medical has undergone significant change in the past three years since investment firm Accelmed Partners acquired the company. With new owners, as well as management and board members, it is no surprise that its CEO and president Adam Szaronos says it feels like a new company.
Szaronos, who joined as chief commercial officer in 2020 before taking on his current role in 2021, tells Optician that Trukera Medical was founded in 2009 under the name TearLab with a focus on osmolarity to solve a unique problem in eye care. Szaronos says in other healthcare settings, the practitioner will review laboratory test results, such as blood, saliva and urine, to inform decision-making. However, he notes, these tests do not exist in eye care because of the unique anatomy of the eye and there not being much to learn about diseases of the eye from these tests.
‘Our company was founded to highlight that ophthalmology and optometry are ripe for objective medical lab tests to help guide decision-making for therapeutic efficacy. So much of what we do in eye care is subjective. We look inside the eye, analyse and try to identify and determine the severity of diseases by looking at it. That’s not done anywhere else.
‘Our founder came up with the idea of taking a 50 nanolitre tear sample and running a rapid lab test looking at biomarkers in the tear. We focused on osmolarity, which is important throughout the body. Your blood is 290-300 milliosmoles per litre and healthy tears should be at the same level. It’s produced from plasma in the blood, but salt is extremely toxic to a cornea.
‘When hyperosmolarity begins to occur, bad things start to happen to the cornea. Our product gave eye care practices around the world the ability to do an instant laboratory test giving objective data right there on the spot. We did that very successfully for a long time. We were the first in the space to do this and, to date, we’ve done that 25 million times around the world in about 50 countries,’ he says.
New opportunities
However, by June 2020, the company had been taken private from the public markets and was looking for opportunities to rebuild, which is when Szaronos joined to lead on a new vision. He explains that TearLab knew it had a strong dry eye asset but opportunities for the test were not in dry eye practices.
‘This test was generating a lot of organic demand in premium vision and surgical practices. These practices weren’t looking for help on diagnosing dry eye. They were looking for a way to identify and flag what we call, in the US at least, our 20/20 unhappy patients. This is where you have talented surgeons and surgical teams that measured the eye, performed great surgery, used all the latest technology to deliver a refractive outcome and, on paper, that outcome looked good – 20/20.
‘But the patient sitting in the post-op chair is saying, “It’s just not as crisp as I thought, do you think maybe something happened with the implant in the eye?” They don’t know how to describe what they’re seeing. They can read 20/20, but it’s not clear there’s an image quality problem, not a refractive error problem.
‘Those surgeons begin looking for a quick way to identify who may have image quality risk factors that can be screened for in advance, educate the patient on and monitor to explain this phenomenon post-operatively if it occurs. Those were the cornea, cataract refractive surgical practices that started using our tests.’
Three years ago, the company was rebranded as Trukera Medical in recognition of its pursuit of 20/20 happy surgical outcomes and a true, healthy cornea. Szaronos says half of Trukera’s product users around the world are optometrists and the other half are ophthalmologists. ‘We think about where the patients are going to be identified prior to a vision surgical event and where that chronic condition needs to be flagged and then managed and tracked over time in our busiest practices,’ he says.
UK priority
Szaronos says the UK is a top priority market for Trukera Medical and it sees growth opportunity as the company switches its thinking about osmolarity as a dry eye diagnosis tool to it complementing premium practices. This shift formed part of the reasoning for the rebrand from TearLab, as well as the old name being associated with the name of the product.
 ‘We were broadening our vision to focus on premium 20/20 happy, but we were also broadening our portfolio. It didn’t make sense to have a company name that was also the name of the product when we were going to start bringing new products to market.
‘We were broadening our vision to focus on premium 20/20 happy, but we were also broadening our portfolio. It didn’t make sense to have a company name that was also the name of the product when we were going to start bringing new products to market.
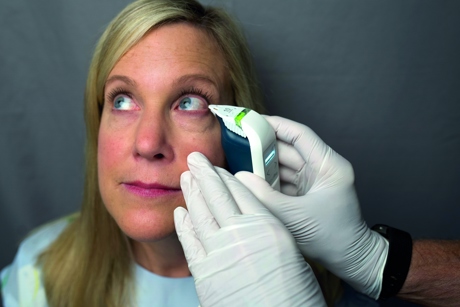
'We needed a company name that multiple products could live beneath. We have the TearLab system, which is our generation one device, and now we’ve launched the ScoutPro osmolarity system (pictured right). Those are the two products beneath the Trukera Medical brand.’
The ScoutPro device reduces the TearLab technology down to the size of a smartphone and offers practitioners portability for osmolarity testing. 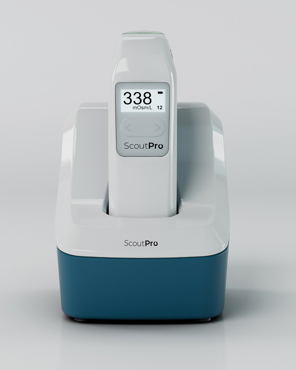 Szaronos says the largest growth area for the company is not necessarily dry eye on its own, but understanding how osmolarity plays a role in the patient journey and why people are incorporating that to help identify and manage dry eye in other areas of their practices.
Szaronos says the largest growth area for the company is not necessarily dry eye on its own, but understanding how osmolarity plays a role in the patient journey and why people are incorporating that to help identify and manage dry eye in other areas of their practices.
‘In the last 10 years, more and more people are understanding the consequences of dry eye, especially how it relates to contact lens wear, Lasik and cataract surgery. If you’re not managing dry eye, you’re missing a disease that compromises vision and quality of life on the most important refractive surface in the entire equation. That’s why we’re seeing all these other subspecialties and other areas start to incorporate dry eye where they haven’t before,’ he says.
Growing demand
Trukera Medical has started to turn its fortunes around and Szaronos shares that it is now cashflow positive and in a position of stability and strength to continue scaling its investment. ‘We’ve changed the pipeline and launched the next gen product. We’ve also started new clinical studies, which are now out in the market and we’re beginning to present these. ‘In the year to date, we’ve launched the Scout Pro in Q1 of this year and we’ve now shipped over 700 systems globally. Our momentum’s building and, for the first time ever, we sold over a hundred osmolarity units just in one month.
‘We’re seeing a huge demand that’s only increasing. It’s not just practices who need help finding dry eye patients. It’s practices saying, “I need to know if there’s a possible leading indicator that this might be a quality of image issue for me because the patient’s about to buy premium contact lenses, or they’re about to pay for Lasik or cataract surgery, and if I miss this, they’re going to blame the investment in the product or the surgery.”’
Szaronos has worked in the eye care industry for 17 years, including 14 years at Alcon. He started in the sales team selling IOLs and cataract surgery equipment then moved into management and led teams across dry eye, surgical glaucoma, vitro-retinal and vision care divisions.
As CEO, he was tasked with leading the turnaround with industry veteran, Jim Mazzo, who joined the board as executive chair. ‘Our vision is to keep building the company around this niche that we’ve created for ourselves and keep addressing the growing demand for the testing space,’ he adds.
Looking ahead, Szaronos teased that the highest growth potential lies outside the US, with the UK a top market where the focus is helping top cataract and refractive surgical customers improve their patient’s quality of vision. ‘We’re a corneal health company and we have our sights set on unmet needs in the broader corneal health space, such as additional diagnostics and therapeutics that tie in nicely with our lab test,’ he says.



